Cause and Prevention
What is Capsular Contracture?
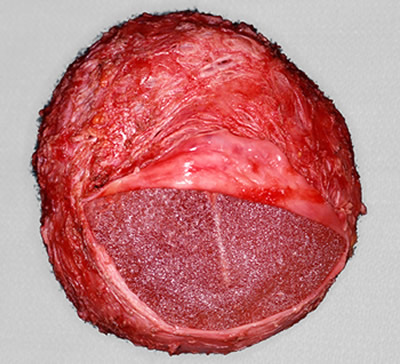
The body normally forms a layer of scar tissue around any implanted foreign object, whether it is a metal artificial joint, plastic chin implant, saline fluid or gel filled breast implant, or an electronic device such as a pacemaker. The scar tissue layer is typically thin and translucent. Capsular contracture refers to a process where the surrounding scar tissue abnormally thickens and contracts tightly around the object within it (Fig. 1).
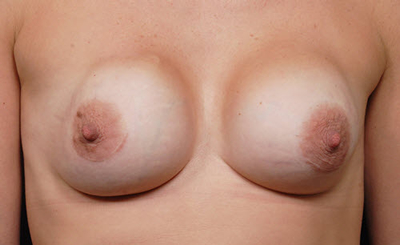
This process does not appear to impact solid objects but saline or silicone filled breast implants become physically compressed. The contractile scar tissue reduces the implant into the smallest possible volume, which is a sphere shape. This makes the breast appear round and distorted in addition to feeling firm (Fig. 2). The contractile process typically pulls the implant upward so that the upper portion of the breast is much more prominent. The breasts become very asymmetric if the opposite side is unaffected.
Capsular contracture causes two problems that drive affected patients to seek treatment: cosmetic deformity and pain. The extent can be quite variable for each component. Plastic surgeons quantify the degree of firmness by the Baker scale: Baker I indicates a completely soft and natural breast consistency with no shape distortion, Baker II a breast that is more firm but still has a normal appearance, Baker III a firm breast with an abnormally round shape, and Baker IV a very firm breast, sometimes rock hard, and a very distorted breast shape (Fig. 2). Breast pain is commonly described as a tightness or chronic discomfort that can be exacerbated by upper extremity and core body movements. However, unlike other chronic pain syndromes such as back pain, medication dependence for relief is unusual.
What Causes Capsular Contracture?
Various theories have been put forth to explain capsular contracture. The infectious theory proposes that bacteria become attached to the implant surface to form a resistant layer called a biofilm. The bacteria within the biofilm produce a protective material called exopolysaccharide that impairs their elimination by antibiotics. Biofilms can be formed by either one or multiple organisms. The most common is Staphylococcus epidermidis. Bacterial biofilm development as the primary cause of capsular contracture is the leading theory and is supported by a growing body of scientific evidence.
There are other reasons for capsular contracture however. Hematomas, a large collection of blood that suddenly develops within the implant pocket due to bleeding, are another inciting cause. Hematomas occur randomly in 1 to 2% of patients soon after surgery. They can also occur up to two weeks from surgery due to the premature resumption of strenuous physical activity.
Neglected, ruptured silicone implants can also lead to capsular contracture. The exposed silicone irritates the surrounding capsule, inciting a chronic inflammatory response that progressively thickens it. Calcifications within the capsule are a byproduct of this process (Fig. 3). Most cases like these are seen in patients who have had implants in place for over 20 to 25 years and have not taken steps to proactively monitor implant condition.
Overtly infected breast implants are fortunately a very rare occurrence but yet another cause of capsular contracture. The infection aspect dominates the immediate treatment focus in these cases with capsular contracture typically developing later.
Patients who smoke have a high incidence of capsular contracture. There are no other known patient risk factors.
Timing and Frequency of Capsular Contracture Onset
Most cases of capsular contracture develop within 6 to12 months of the original surgical procedure. However, capsular contracture can occur many years later without apparent antecedent cause. Most cases affect only one side. Double capsules (i.e. both sides affected), while less common, usually represent a more recalcitrant condition.
The overall incidence of capsular contracture varies widely in the surgical literature and is higher in breast reconstruction cases compared to cosmetic breast augmentation. While less common today the incidence is still about 5% for breast augmentation, although a few studies report even less.
Capsules can occur with either saline or silicone implants but are slightly less frequent with the former. Breast implants placed beneath the pectoral muscle are associated with a decreased incidence of capsular contracture. The implant surface can be either smooth or textured (rough). It was once believed that textured implants decrease the incidence of capsular contracture. However later studies have shown the difference between the two types is small as long as they are placed under and not in front of the pectoral muscle. Textured implants have recently been associated with an extremely rare form of lymphoma, so many surgeons avoid them altogether today.
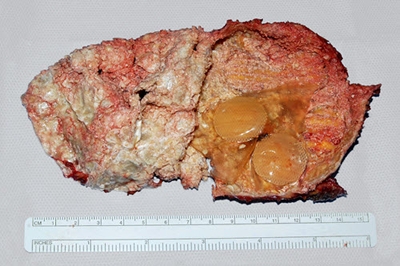
Figure 3. The inside surface of an implant capsule that has been opened and separated from the ruptured implant is shown. This is a particularly severe case. The white textured areas represent calcium deposits. Remnants of the implant cover with attachment patches (used in the first generation implants) are seen to the bottom right
Strategies for Capsular Contracture Prevention
Strict sterile technique is a prerequisite for the prevention of capsular contracture but is not by itself infallible. Cardinal principles that include implant placement under the pectoral muscle and making sure that all exposed blood vessels have been meticulously cauterized are essential. Preoperative intravenous antibiotics have been shown to be the most effective method of antibiotic administration. Most plastic surgeons continue oral antibiotics postoperatively as an added (although unproven) insurance measure. Selecting saline instead of silicone implants can further contribute to a decreased capsular contracture rate. However the latter type are much more popular with patients for other reasons. There is also some recent evidence that incisions in the breast crease are better than incisions around the areola with respect to preventing capsular contracture.
There are additional specific intraoperative measures that contribute to a decreased capsular contracture rate. These include all scrubbed personnel changing gloves immediately before implant placement, opening the breast implant boxes just prior to insertion, minimal handling of the devices both before and after insertion, irrigating the implants and the implant pockets with an antibiotic solution, isolating the skin using a sterile plastic barrier, and the optional use of a plastic funnel to further decrease implant contact with the skin during insertion.
Using certain preventative medications after surgery may be helpful (see Treatment Options). However, the results of this practice have been mixed so far.
Patients who smoke must stop completely (and forever) if they wish to eliminate this major risk factor. Cutting back alone is not effective.
Postoperative implant massage is 1970’s technology without any formal scientific evidence to prove its efficacy. It may even contribute to prolonged inflammation. It is no longer uniformly recommended by plastic surgeons today.